
In modern steelmaking, the slag is far more than a protective layer floating on molten steel — it is a reactive chemical reactor that governs sulfur removal, inclusion absorption, and reoxidation prevention. The single most important parameter controlling slag performance is basicity, typically expressed as the ratio of basic oxides (CaO, MgO) to acidic oxides (SiO₂, P₂O₅). A deep understanding of slag chemistry allows metallurgists to engineer slags that maximize desulfurization while minimizing refractory wear and inclusion-related defects.
This article explores the fundamental chemistry of steelmaking slags, their interaction with deoxidizing alloys, and practical strategies for optimizing basicity across different steel grades and process routes.
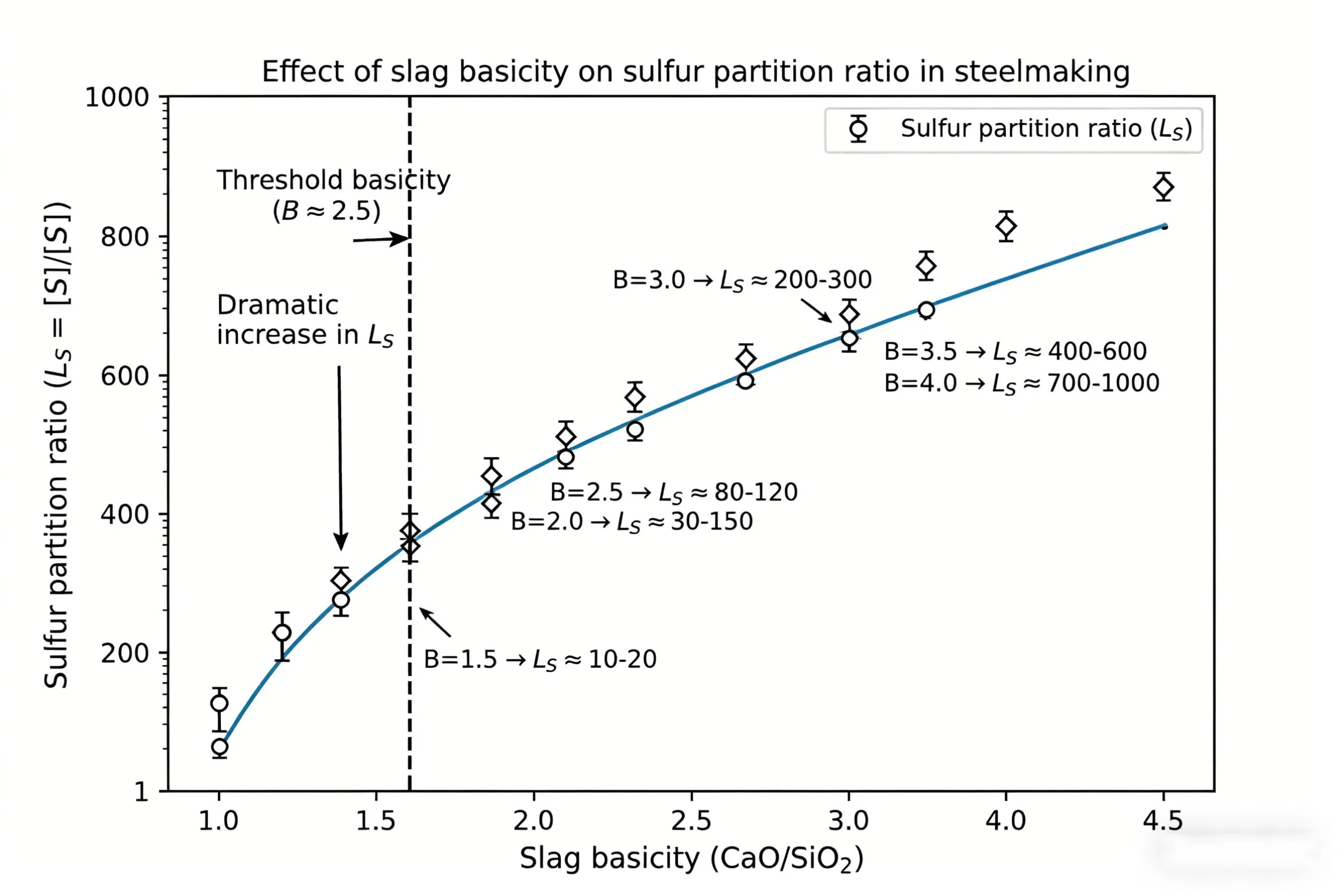
What Is Slag Basicity? Defining the CaO/SiO₂ Ratio
Basicity (B) is most commonly expressed as the mass ratio of CaO to SiO₂ in the slag. This ratio determines the availability of free oxygen ions (O²⁻) in the slag melt, which directly drives desulfurization and dephosphorization reactions. Slags are classified as:
- Acidic slags (B < 1.0): High SiO₂, low CaO. Poor desulfurization, but less aggressive on acidic refractories. Rarely used in modern ladle refining.
- Neutral slags (B = 1.0–2.0): Moderate desulfurization capability. Sometimes used for certain carbon steel grades.
- Basic slags (B > 2.0): High CaO availability. Excellent desulfurization and inclusion absorption. Standard for clean steel production.
For most clean steel applications, target basicity ranges from 2.5 to 4.5, with higher values reserved for ultra-low-sulfur grades (e.g., pipeline, bearing, and automotive AHSS).
The Desulfurization Reaction: How Basicity Drives Sulfur Removal
Sulfur is removed from steel via the slag-metal reaction. The overall desulfurization reaction can be written as:
[S] + (O²⁻) → (S²⁻) + [O]
The free oxygen ions (O²⁻) are supplied by basic oxides, primarily CaO. The sulfur partition ratio (Ls = [%S]slag / [%S]steel) increases exponentially with slag basicity. Empirical data show:
- At B = 1.5, Ls ≈ 20–50 → final sulfur 0.015–0.030%
- At B = 2.5, Ls ≈ 80–150 → final sulfur 0.008–0.015%
- At B = 3.5, Ls ≈ 200–400 → final sulfur 0.003–0.008%
However, basicity alone is insufficient. Low slag FeO (below 1%) and high slag fluidity are equally critical for rapid sulfur transport to the slag-metal interface.
Slag-Inclusion Interaction: Absorbing Deoxidation Products
When deoxidizers like aluminum or silicon-manganese are added, they form oxide inclusions (Al₂O₃, MnO·SiO₂). These inclusions must be absorbed by the slag to prevent their entrapment in the solidifying steel. Slag basicity determines inclusion absorption capacity and the resulting inclusion chemistry.
Alumina (Al₂O₃) absorption: Highly basic slags (B > 3.0) dissolve alumina rapidly, forming calcium aluminates in the slag. The absorption capacity follows: CaO-rich slags can hold up to 30–40% Al₂O₃ before saturation, whereas acidic slogs quickly become saturated, leaving alumina inclusions in the steel.
For silicon-manganese deoxidation: The resulting MnO·SiO₂ inclusions are liquid and more easily absorbed, but basic slags still outperform acidic slags in overall inclusion removal. Maintaining a basic slag also prevents reversion of sulfur and phosphorus from the slag back into the steel.
Optimizing Basicity Across Steel Grades
Different steel grades demand different slag basicity targets. Below is a practical guide:
| Steel Grade | Target Basicity (CaO/SiO₂) | Key Objectives | Typical Final Sulfur (ppm) |
|---|---|---|---|
| Construction / Rebar | 1.8–2.5 | Basic desulfurization, cost efficiency | 150–300 |
| Structural / HSLA | 2.5–3.5 | Good desulfurization + inclusion control | 50–120 |
| Automotive AHSS / DP steel | 3.0–4.0 | Low S, clean inclusions for formability | 20–50 |
| Pipeline (API X70+) | 3.5–4.5 | Ultra-low S for HIC resistance | <15 |
| Bearing / Spring steel | 3.5–4.5 | Maximum cleanliness, fatigue life | <10 |
Practical Strategies for Basicity Control
Achieving and maintaining target basicity requires systematic slag engineering. Key practices include:
- Ladle slag carryover control: Minimize BOF/EAF slag carryover during tapping (target < 5 kg/ton). Oxidizing slags with high FeO will consume deoxidizers and reduce basicity.
- Top slag addition: Add lime (CaO) and synthetic refining fluxes to achieve target basicity. For every 1% increase in CaO, basicity rises by approximately 0.3–0.5 units depending on SiO₂ level.
- Aluminum addition: Al deoxidation reduces slag FeO and indirectly increases effective basicity by lowering oxidizing potential.
- Fluidity optimization: Add fluorspar (CaF₂) or alumina to adjust slag viscosity at high basicity — excessively viscous slags hinder sulfur mass transfer.
- Real-time monitoring: Use XRF or portable slag analyzers to verify basicity during ladle treatment; adjust lime additions accordingly.

The Trade-Off: Basicity vs. Refractory Life
Highly basic slags (B > 4.0) are corrosive to MgO-C and MgO-spinel ladle refractories. The chemical reaction: MgO(s) + CaO·SiO₂(l) forms low-melting-point magnesium silicates, accelerating wear. To balance refractory life and metallurgical performance:
- For routine grades, maintain B = 2.5–3.0 — adequate desulfurization with moderate refractory wear.
- For ultra-low-sulfur grades, use short treatment times and consider MgO-saturated slags (add dolomitic lime) to reduce MgO dissolution.
- Apply slag splashing after tapping to coat refractories with a protective basic layer.
Case Study: Pipeline Steel Slag Optimization
A steel mill producing API X70 linepipe experienced inconsistent sulfur levels (25–60 ppm) and occasional hydrogen-induced cracking (HIC) failures. Initial slag basicity varied between 2.0 and 3.2 due to inconsistent lime addition and BOF slag carryover. After implementing a targeted slag engineering protocol — limiting carryover to 4 kg/ton, adding 8 kg/ton of high-CaO synthetic slag, and maintaining B = 3.8–4.2 — sulfur levels stabilized below 12 ppm. HIC testing passed with zero cracks, and refractory life decreased by only 8%, an acceptable trade-off for the quality improvement.
Slag basicity optimization is not merely a chemistry exercise — it is a strategic lever connecting deoxidation practice, inclusion engineering, sulfur removal, and refractory management. By understanding the interplay between CaO/SiO₂ ratio, sulfur partition, and inclusion absorption, steelmakers can consistently produce cleaner, tougher, and more reliable steel. Bright Alloys provides high-purity ferrosilicon, silicon-manganese, and synthetic slag additives to support every aspect of modern ladle metallurgy.