
Oxygen is both essential and detrimental in steelmaking. While oxygen blowing is fundamental to basic oxygen furnace (BOF) and electric arc furnace (EAF) refining to remove carbon, phosphorus, and silicon, dissolved oxygen remaining after tapping must be rigorously controlled. Uncontrolled oxygen leads to gas porosity, embrittlement, and most critically, the formation of non-metallic inclusions that compromise mechanical properties, fatigue life, and surface quality.
Modern steelmaking demands an intimate understanding of oxygen activity, deoxidation equilibria, and inclusion engineering. This article examines how dissolved oxygen drives inclusion formation and presents practical strategies for optimal deoxidation practice across various steel grades.
The Oxygen Challenge: From BOF to Tundish
At the end of the BOF or EAF process, molten steel contains 400–800 ppm dissolved oxygen, primarily in equilibrium with carbon. For reference, most finished steel products require oxygen levels below 30 ppm, with critical applications (bearing steels, spring steels) demanding less than 10 ppm total oxygen. The deoxidation step — adding elements with high oxygen affinity — must remove the vast majority of this oxygen while managing the inevitable inclusion population that results.
The fundamental deoxidation reaction can be represented as: x[M] + y[O] → MₓOy (s or l). The choice of deoxidizer determines inclusion chemistry, morphology, and removal behavior. Let’s examine the most common deoxidation systems.
Aluminum Deoxidation: High Efficiency, High Inclusion Risk
Aluminum is the most powerful and cost-effective deoxidizer, capable of reducing dissolved oxygen to 2–5 ppm under equilibrium conditions. The reaction is: 2Al + 3[O] → Al₂O₃(s). However, the resulting alumina (Al₂O₃) inclusions are solid, hard, and often form clusters that are difficult to remove completely. These inclusions are detrimental to fatigue life, machinability, and surface finish. In continuous casting, alumina buildup in submerged entry nozzles (SEN) is a persistent operational challenge.

Best practice for Al deoxidation: For fully killed steels requiring very low total oxygen, use 0.5–1.2 kg Al per ton of steel. Follow with argon stirring to promote inclusion flotation and, where possible, calcium treatment to modify alumina into liquid calcium aluminates.
Silicon-Manganese Deoxidation: Liquid Inclusions, Cleaner Steel
The combination of silicon and manganese offers a distinct advantage: the deoxidation product is a liquid manganese silicate (MnO·SiO₂) at steelmaking temperatures. Liquid inclusions coalesce more readily and float out faster than solid oxides. The reaction follows: [Si] + 2[Mn] + 4[O] → (MnO)₂·SiO₂(l). While silicon-manganese deoxidation does not achieve as low oxygen levels as aluminum (typically 20–40 ppm residual O), the resulting inclusion population is smaller, more spherical, and less harmful. For many structural steel grades, Si-Mn deoxidation provides the optimal balance between cleanliness and cost.
Practical tip: Maintain a target Mn/Si ratio of 3:1 to 5:1 to ensure liquid oxide formation. Use high-quality silicon-manganese alloy (SiMn) with consistent chemistry for reproducible results.
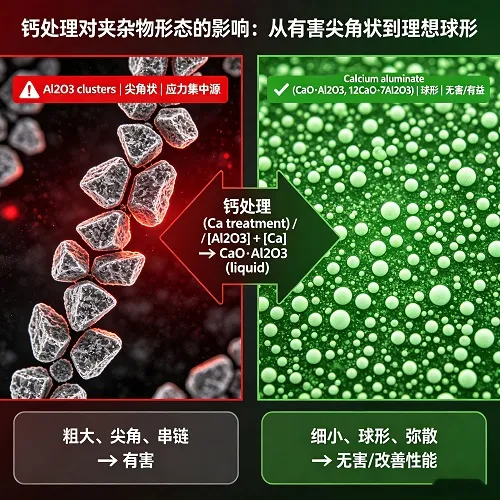
Calcium Treatment: Modifying Inclusions for Superior Performance
Calcium is rarely used as a primary deoxidizer due to its high cost and low recovery, but it is unmatched as an inclusion modifier. When added to aluminum-killed steel (typically via CaSi cored wire), calcium reacts with solid alumina inclusions to form low-melting-point calcium aluminates (e.g., 12CaO·7Al₂O₃, melting point ~1455°C). These globular inclusions are less harmful to mechanical properties and significantly reduce nozzle clogging during continuous casting.
Calcium addition guidelines: For optimal modification, aim for a Ca/Al ratio of 0.10–0.15. Excessive calcium leads to CaS formation, which can re-solidify and cause other casting issues. Precision is critical; modern practice uses CaSi cored wire injection with real-time feedback.
Measuring and Monitoring Oxygen Activity
Modern steelmaking relies on electrochemical oxygen sensors (ZrO₂-based) to measure dissolved oxygen activity directly in the ladle. These measurements guide deoxidant additions, reducing over-treatment and under-treatment. Key oxygen targets by process stage:
- End of BOF/EAF: 400–800 ppm (before deoxidation)
- After Al or SiMn addition: 10–30 ppm (active oxygen)
- After calcium treatment: 5–15 ppm + stable inclusion modification
- Tundish (continuous casting): Total oxygen (Otot) typically 15–30 ppm, depending on grade
Strategies for Optimal Deoxidation Practice
Achieving consistent steel cleanliness requires a systematic approach. The following framework applies across most carbon and low-alloy steel grades:
- Slag engineering: Maintain basic slag (CaO/SiO₂ > 2.5) to absorb deoxidation products. Reducing slag FeO below 1% minimizes oxygen reversion.
- Strong argon stirring: At least 5–10 minutes of gentle argon bubbling after deoxidation promotes inclusion flotation.
- Sequential addition: For grades requiring very low oxygen, consider pre-deoxidation with Si-Mn followed by Al finishing, then Ca modification.
- Ladle covering: Prevent reoxidation from ladle slag or air entrainment during tapping and casting.
Case Example: Bearing Steel Quality Transformation
A specialty steel producer manufacturing SAE 52100 bearing steel faced high rejection rates due to alumina-type inclusions detected in ultrasonic testing. By implementing a two-step deoxidation protocol (Si-Mn pre-deoxidation → Al finishing → CaSi cored wire treatment) and optimizing ladle stirring time to 12 minutes, the producer reduced total oxygen from 18 ppm to 8 ppm. Inclusion ratings improved by 60%, and bearing fatigue life (L10) more than doubled. This case underscores that oxygen control is not a single action but an integrated process strategy.
As steel applications demand ever-higher performance — from electric vehicle drivetrains to offshore wind foundations — mastery of oxygen control becomes a competitive differentiator. By understanding the relationship between dissolved oxygen, inclusion formation, and deoxidation chemistry, steelmakers can consistently produce cleaner, stronger, and more reliable steel. Bright Alloys offers a full portfolio of deoxidation alloys, including ferrosilicon, silicon-manganese, and CaSi cored wire, backed by metallurgical expertise to help optimize your practice.